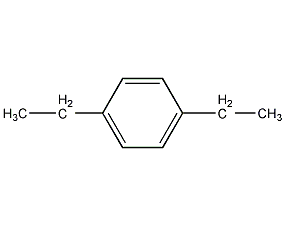
Structural formula
| Business number | 02R7 |
|---|---|
| Molecular formula | C10H14 |
| Molecular weight | 134.22 |
| label |
1,4-diethylbenzene, 1,4-Diethylbenzene, desorbent, Cross-linking agent for polystyrene resin, high boiling point solvents, Aromatic hydrocarbons |
Numbering system
CAS number:105-05-5
MDL number:MFCD00009264
EINECS number:203-265-2
RTECS number:CZ5640700
BRN number:1903396
PubChem number:24859254
Physical property data
1. Properties: colorless liquid[1]
2. Melting point (℃): -42.8[2]
3. Boiling point (℃): 183.7[3]
4. Relative density (water=1): 0.86[ 4]
5. Relative vapor density (air=1): 4.6[5]
6. Saturated vapor pressure (kPa) : 1.33 (62.8℃)[6]
7. Critical pressure (MPa): 2.8[7]
8 .Octanol/water partition coefficient: 4.45[8]
9. Flash point (℃): 55 (CC)[9]
10. Ignition temperature (℃): 430[10]
11. Explosion upper limit (%): 6.0[11]
12. Lower explosion limit (%): 0.7[12]
13. Solubility: insoluble in water, soluble in ethanol, benzene, tetracycline Most organic solvents such as carbon chloride, ether, acetone, etc. [13]
14. Flash point (ºC): 430
15. Heat of evaporation (KJ/mol, 25ºC): 52.50
16. Heat of fusion (KJ/mol): 10.59
17. Heat of formation (KJ/mol, 298K, gas): 22.27
18. Specific heat capacity (KJ/(kg ·K), 298K, constant pressure): 1.31
Toxicological data
1. Acute toxicity[14] LD50: 1200mg/kg (rat oral)
2. Irritation No data available
3. Subacute and chronic toxicity[15] Rats, guinea pigs, rabbits and monkeys inhaled 2.61~9.5g/m3 for 7 hours a day for a total of 103~138 days. Mild pathological changes were found in the liver, kidneys and testicles.
Ecological data
1. Ecotoxicity[16] LC50: 2.49mg/L (96h) (medaka)
2. Biodegradability No information available
3. Non-biodegradability[17] In the air, when the concentration of hydroxyl radicals is 5.00×105/cm3, the degradation half-life is 2d (theoretical).
4. Bioconcentration[18] BCF: 530 (theoretical): 362~598 (carp, Exposure concentration 20mg/L, exposure time 6 weeks); 320~629 (carp, exposure concentration 2mg/L, exposure time 6 weeks)
5. Other harmful effects [19] This substance is harmful to the environment and harmful to aquatic biological systems.�Give special attention. Bioaccumulation occurs especially in fish. But once out of the polluted water body, the pollutants in the fish are discharged relatively quickly.
Molecular structure data
1. Molar refractive index: 45.35
2. Molar volume (cm3/mol): 155.0
3. Isotonic specific volume (90.2K ): 360.4
4. Surface tension (dyne/cm): 29.1
5. Dielectric constant: 2.34
6. Dipole moment (10-24cm3):
7. Polarizability: 17.97
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): 3.5
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 0
4. Number of rotatable chemical bonds: 2
5. Number of tautomers: none
6. Topological molecule polar surface area 0
7. Number of heavy atoms: 10
8. Surface charge: 0
9. Complexity: 66
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. Stability[20] Stable
2. Incompatible substances [21] Strong oxidants, acids, halogens, etc.
3. Polymerization hazards[22] No aggregation
Storage method
Storage Precautions[23] Store in a cool, ventilated warehouse. The storage temperature should not exceed 37℃. Keep away from fire and heat sources. Keep container tightly sealed. should be kept away from oxidizer, do not store together. Use explosion-proof lighting and ventilation facilities. It is prohibited to use mechanical equipment and tools that are prone to sparks. The storage area should be equipped with emergency release equipment and suitable containment materials.
Synthesis method
None
Purpose
1. Mainly used for solvents, organic synthesis, and gas chromatography comparison samples.
2. Used as solvent and organic synthesis intermediate. [24]

 微信扫一扫打赏
微信扫一扫打赏

