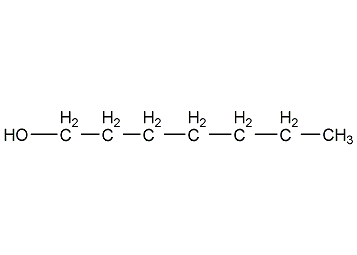
Structural formula
| Business number | 0344 |
|---|---|
| Molecular formula | C7H16O |
| Molecular weight | 116.20 |
| label |
n-Heptanol, Heptanol, n-Heptyl alcohol, 1-Hydroxyheptane, Enanthic alcohol, synthetic fragrances, solvents for cosmetics, alcohol solvent |
Numbering system
CAS number:111-70-6
MDL number:MFCD00002986
EINECS number:203-897-9
RTECS number:MC0350000
BRN number:1731686
PubChem number:24886275
Physical property data
1. Properties: Colorless viscous liquid with strong aromatic odor.
2. Relative density (g/mL, 20/4℃): 0.8219
3. Melting point (ºC): -33.2
4. Boiling point ( ºC, 101.3kPa): 176.45
5. Refractive index (n20ºC): 1.4249
6. Flash point (ºC, closed): 73
7. Heat of evaporation (KJ/mol): 213.52
8. Heat of generation (KJ/mol): -406.92
9. Heat of combustion (KJ/mol): 4635.2
10. Vapor pressure (kPa, 42.4ºC): 0.13
11. Heat of combustion (KJ/mol): 4618.5
12. Critical temperature (ºC): 365
13. Thermal conductivity (W/(m·K), 20ºC): 16.33
14. Freezing point (ºC): -35
15. Dissolution Property: Slightly soluble in water, miscible with general organic solvents such as alcohol and ether. 1.0 g of 1-heptanol can be dissolved in 1L at 18°C, and the aqueous solution is in the form of a colloid.
16. Relative density (25℃, 4℃): 0.8187
17. Refractive index at room temperature (n25): 1.4223
18. Critical pressure (MPa): 3.058
19. Critical density (g·cm-3): 0.267
20. Critical volume ( cm3·mol-1): 435
21. Critical compression factor: 0.253
22. Eccentricity factor: 0.587
23. Solubility parameter (J·cm-3)0.5: 21.517
24. van der Waals area ( cm2·mol-1): 1.168×1010
25. van der Waals volume (cm3·mol-1): 83.090
26. Gas phase standard heat of combustion (enthalpy) (kJ·mol-1) : 4704.62
27. Gas phase standard claimed heat (enthalpy) (kJ·mol-1): -336.56
28. Gas phase standard entropy (J· mol-1·K-1): 480.3
29. Gas phase standard formation free energy (kJ·mol-1):-125.81
30. Liquid phase standard combustion heat (enthalpy) (kJ·mol-1): -4637.8
31. Liquid phase standard claimed heat (enthalpy) ( kJ·mol-1): -403.4
32. Liquid phase standard entropy (J·mol-1·K-1): 320.1
33. Liquid phase standard formation free energy (kJ·mol-1): -144.85
34. Liquid phase standard Hot melt (J·mol-1·K-1): 262.6
Toxicological data
1. Acute toxicity: Oral LD50 in rats: 500 mg/kg; Inhalation LC50 in mice: 6600mg/m3, 2 hours
2. It is of low toxicity. The toxicity is similar to that of hexanol. The olfactory threshold concentration is 0.27mg/m3. The maximum allowable concentration in the workplace is 474mg/m3
Ecological data
This substance is not harmful to water bodies.
Molecular structure data
1. Molar refractive index: 36.01
2. Molar volume (cm3/mol): 141.5
3. Isotonic specific volume (90.2K ): 327.3
4. Surface tension (dyne/cm): 28.5
5. Polarizability (10-24cm3): 14.27
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): None
2. Number of hydrogen bond donors: 1
3. Number of hydrogen bond acceptors: 1
4. Number of rotatable chemical bonds: 5
5. Number of tautomers: none
6. Topological molecule polar surface area 20.2
7. Number of heavy atoms: 8
8. Surface charge: 0
9. Complexity: 35.4
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 0
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. Prohibit contact with strong oxidants, acid chlorides, and acid anhydrides.
2. Colorless liquid. This ingredient is found in some essential oils such as hyacinth and violet. Heptan alcohol has a bright green fruity aroma with a winey waxy aroma, and the aroma is like sweet wine, nuts, and green. It is insoluble in water, soluble in most organic solvents, and soluble in 70% ethanol (1:1) . Flammable liquid, non-corrosive to metals, low vapor pressure, safer to use. Chemically reactive with primary alcohols.
3. Exist in tobacco leaves.
Storage method
Store in a cool, ventilated warehouse. Keep away from fire and heat sources. They should be stored separately from oxidants and food chemicals, and avoid mixed storage. Equipped with the appropriate variety and quantity of fire equipment. The storage area should be equipped with emergency release equipment and suitable containment materials. Safely stored in iron, mild steel, copper or aluminum containers.
Synthesis method
1. Obtained from the reduction of n-heptaldehyde. Stir 1.8kg iron filings, 3L water and 0.45kg heptaldehyde together, heat to reflux for 6-7 hours, and then perform steam distillation to obtain approximately 7-8L of distillate. Separate the oil layer and add 1L 20% heptyl hydroxide for hydrolysis. . Separate the oil layer for distillation, collect the 172-176°C fraction, and obtain about 350g heptanol. The reduction reaction can also be carried out in the presence of dilute hydrochloric acid. In addition, heptanol can also be produced by reacting pentane with ethylene oxide in the presence of anhydrous aluminum bromide.
2. Refining method: Shake repeatedly with alkaline potassium permanganate solution until the color of potassium permanganate does not disappear within 15 minutes, then dry with potassium carbonate or calcium oxide and fractionate.
3. Tobacco: FC, 18; FC, 40. It is prepared by reducing heptanal with iron filings in dilute acetic acid.
4. Preparation method:
![]()
In a 12L reaction bottle equipped with a stirrer and reflux condenser, add 1800g (32.2mol) iron filings, 3000mL (52.5mol) glacial acetic acid, 3000mL water and 450g (3.95mol) heptanal (2) , react in a steam bath with stirring for 6 to 7 hours. Install the steam distillation device and steam distill until no oil is distilled, and collect about 7 to 8L of distilled liquid. Separate the organic layer. The aqueous layer is distilled to recover a small amount of heptanol-1. Combine the obtained products, add 1000 mL (20%) sodium hydroxide solution, and stir in a steam bath for 4 hours to hydrolyze n-heptyl acetate. Separate the oil layer, distill it, and collect the fraction between 172 and 176°C. Add 100mL of water to the residue in the flask, and then distill. A small amount of oil will distill out along with the water vapor. Mix it with the fraction obtained above, separate the water, and then distill. Collect the oil at 172-176°C. Fraction, 350-370g of product (1) is obtained, with a yield of 75%-81%. [1]
5. Preparation method:
![]()
Into a 20L reaction bottle equipped with a stirrer and reflux condenser, add 1.8kg (32.2mol) of iron filings, 3L of glacial acetic acid, 3L of water, and 450g (3.95mol) of heptanal (2), while stirring. React in steam bath for 6 to 7 hours. Then carry out steam distillation until no more oil droplets distill out, and collect about 7 to 8 L of distillate. Separate the oil layer. The water layer is then distilled until no oil is distilled out, and the oil layer is separated. Combine the oil layers, add 1000 mL (20%) sodium hydroxide solution, and stir in a steam bath for 4 hours to hydrolyze heptyl acetate in it. After cooling, separate the oil layer, distill it, and collect the fraction between 172 and 176°C. Add water to the residue and then perform steam distillation to recover part of the product. The total product 1-heptanol①(1) was 350-370g, with a yield of 75%-81%. Note: ① Zinc powder can also be used instead of iron filings for reduction. [2]
Purpose
1. Heptanol has been widely used for only 50 years. Its application is limited by one of its greasy and pungent properties. The dosage of several types of edible flavors is limited to 0.9 to 5 mg/kg. The application of daily chemical fragrances: soap fragrance 0.01% to 0.1%; detergent 0.001% to 0.01%; perfume fragrance 0.05% to 0.1%; cream fragrance 0.005% to 0.03%.
It is a synthetic raw material for dianthus and jasmine-flavored spices, a solvent for cosmetics and an organic synthetic raw material.
2.Can be used for food flavors, the dosage of food flavors is 0.9~5mg/kg. Used as soap flavor, detergent flavor, perfume flavor, cream flavor, etc.
Distill and collect the fractions between 172 and 176°C. Add water to the residue and then perform steam distillation to recover part of the product. The total product 1-heptanol①(1) was 350-370g, with a yield of 75%-81%. Note: ① Zinc powder can also be used instead of iron filings for reduction. [2]
Purpose
1. Heptanol has been widely used for only 50 years. Its application is limited by one of its greasy and pungent properties. The dosage of several types of edible flavors is limited to 0.9 to 5 mg/kg. The application of daily chemical fragrances: soap fragrance 0.01% to 0.1%; detergent 0.001% to 0.01%; perfume fragrance 0.05% to 0.1%; cream fragrance 0.005% to 0.03%.
It is a synthetic raw material for dianthus and jasmine-flavored spices, a solvent for cosmetics and an organic synthetic raw material.
2.Can be used for food flavors, the dosage of food flavors is 0.9~5mg/kg. Used as soap flavor, detergent flavor, perfume flavor, cream flavor, etc.

 微信扫一扫打赏
微信扫一扫打赏

